AI Software Development Agency: How to Choose the Right Partner
The AI Software Development Market in 2026 £337.75bn UK AI market size by 2032 (26.4% CAGR) £800–£1,500+ Daily rates for mid-market...
20 min read
 Peter Vogel
:
Updated on March 27, 2026
Peter Vogel
:
Updated on March 27, 2026

The pharmaceutical and biotechnology sectors across the United Kingdom stand at a critical inflection point. Whilst traditional drug discovery pipelines continue to consume an average of 10 to 12 years and approximately £2.3 to £2.6 billion per approved drug, artificial intelligence platforms are demonstrating the capacity to compress discovery timelines by 40 to 80 percent in early-stage research phases. For UK pharma and biotech leaders seeking to maintain competitive positioning within an increasingly AI-driven global landscape, understanding the practical applications, regulatory frameworks, and implementation pathways for AI in drug discovery is no longer discretionary. This article examines how UK pharmaceutical and biotech companies are accelerating new treatments through artificial intelligence, the evidence supporting these advances, and the strategic frameworks for successful implementation.
The UK biotechnology market reached USD 22.00 billion in 2024 and is projected to expand to USD 41.47 billion by 2033, representing a compound annual growth rate of 7.22 percent. This expansion is substantially driven by artificial intelligence integration across research and development operations. The global market for AI in pharma and biotech is expanding far more rapidly, projected to grow from USD 8.54 billion in 2026 to USD 154.10 billion by 2034, representing a compound annual growth rate of 43.55 percent. European markets are anticipated to expand at 44.70 percent over the same period, with the UK accounting for approximately 4.7 percent of global AI pharma revenues as of 2026—a figure indicating substantial growth potential and current under-penetration compared to North America, which captured 51.73 percent of global market share in 2025.
The UK government has positioned AI adoption as a cornerstone of the national life sciences strategy. The government's "AI for Science Strategy," published in November 2025, articulates an ambitious objective: achieving trial-ready drugs within 100 days by 2030. UK Research and Innovation (UKRI) announced more than GBP 1 billion in quantum computing funding over four years as of December 2025, with over GBP 500 million dedicated specifically to quantum computing applications in sectors including pharmaceuticals. Additionally, the government committed GBP 47.8 million through the Voluntary Branded Medicines Pricing, Access and Growth (VPAG) Investment Programme, aimed at channelling approximately GBP 400 million of investment into the UK's health and life sciences sector by 2028.
Evidence of AI adoption across the UK pharmaceutical and biotech sectors indicates rapid integration into research workflows. As of 2025, 87 percent of life sciences researchers now use AI for work-related research tasks, up from 75 percent in 2023. Over 90 percent of life sciences manufacturers reported that they were using or adopting smart technology as of 2024. A survey of UK life sciences chief executive officers conducted by KPMG identified that 64 percent were prioritising AI investments, indicating broad recognition of AI's strategic importance at leadership level across the sector.
Leading UK pharmaceutical companies have invested substantially in workforce capability development around artificial intelligence. AstraZeneca, ranked as the leading UK pharmaceutical innovator, completed generative AI certifications for approximately 12,000 employees as of April 2025, with 93 percent of trained staff reporting positive impact on their work. This level of organisational commitment to AI literacy represents a significant investment in human capital and suggests that leading UK pharma companies view AI integration as a fundamental operational transformation rather than a discrete technology project.
However, many organisations remain stuck in pilot mode and unable to scale AI across global operations whilst managing regulatory complexity and patient data sensitivity. Successful organisations embed AI as a layer within previously established deterministic workflows rather than replacing them entirely, thereby creating hybrid systems that combine AI predictions with traditional safety and compliance frameworks.
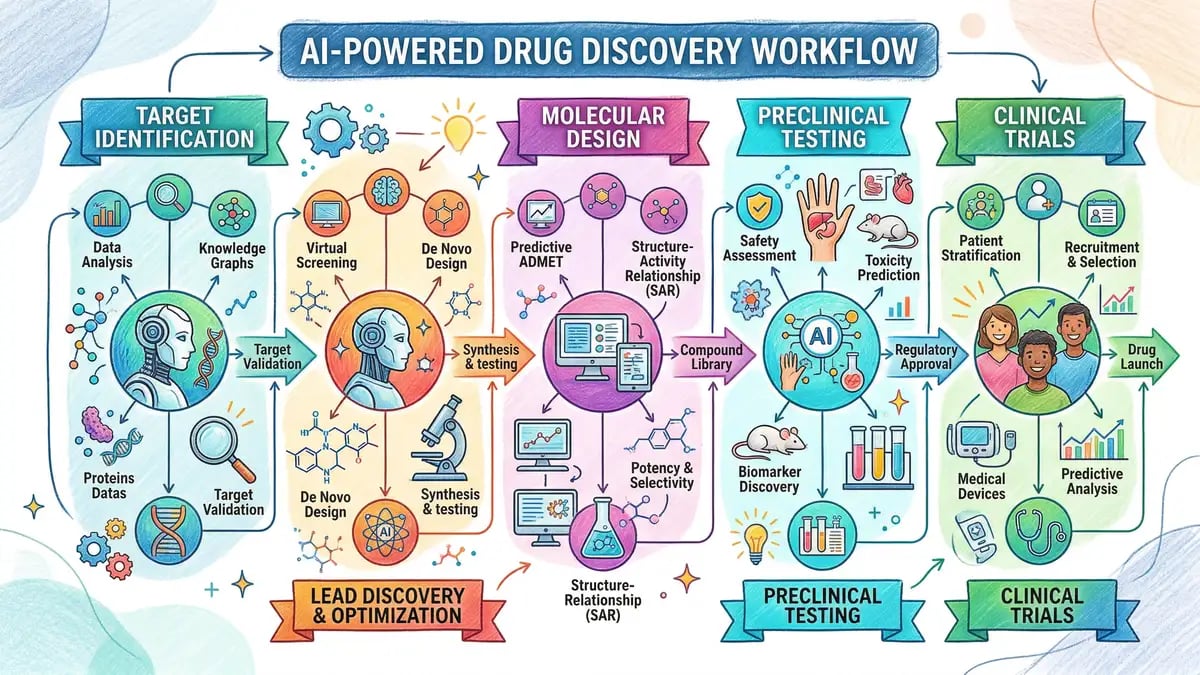
Artificial intelligence demonstrates differentiated value across distinct phases of pharmaceutical development. The highest-impact applications cluster in early discovery phases and clinical trial optimisation, where traditional approaches are characterised by high cost, lengthy timelines, and substantial attrition.
The earliest phase of drug discovery, target identification, represents perhaps the most mature and demonstrably valuable application of artificial intelligence in the pharmaceutical pipeline. Computational approaches to target discovery have evolved from simple database queries to sophisticated machine learning models capable of integrating multi-omics datasets spanning genomics, transcriptomics, proteomics, metabolomics, and imaging data. Sanofi deployed AI-driven "Target Discovery engines" that generated seven new therapeutic targets within a single year, substantially accelerating the pace of novel target identification compared to traditional approaches.
Deep learning models, particularly graph neural networks and transformer-based architectures, have demonstrated particular utility in integrating heterogeneous biological data to prioritise targets based on their biological relevance, therapeutic potential, and probability of successful modulation. Recent research employing autoencoder-based frameworks for multi-omics integration in oncology identified not only established therapeutic targets but also six novel genetic targets, demonstrating the capacity of artificial intelligence to uncover cryptic therapeutic opportunities that would remain hidden within traditional single-data-modality approaches.
The phase of molecular design and lead optimisation historically represents one of the most time-consuming and resource-intensive stages of drug development, often requiring hundreds of synthetic cycles and biological assays to identify compounds with optimal balance of potency, selectivity, solubility, and safety profile. Generative artificial intelligence models have transformed this phase by enabling the creation of novel compounds based on learned patterns from chemistry and assay data, moving from screening of known libraries to design of previously unknown molecules with predicted optimised pharmacological profiles.
Merck's KERMT model, developed in collaboration with NVIDIA and trained on more than 11 million molecules, exemplifies the application of foundation models to small-molecule drug discovery. By predicting molecular behaviour in the body and identifying potential safety issues at the preclinical stage, KERMT reduces the need for months of laboratory work and enables teams to compress timelines by approximately 30 percent. The model is made available as open-source software, allowing the broader scientific community to leverage advances in deep learning for molecular design. According to Alan Cheng, Senior Director of Data Science at Merck, AI is "speeding up the early stages of drug development dramatically, sometimes cutting timelines by 30% or more, improving drug candidate quality and reducing costs."
The phase of clinical trial design and execution represents the most costly phase of drug development, with Phase III trials alone consuming 40 to 50 percent of total clinical development budgets and late-stage failures capable of costing upwards of USD 1 billion. Artificial intelligence applications in clinical development focus on protocol optimisation, patient recruitment and stratification, real-time safety monitoring, and integration of real-world evidence to supplement traditional clinical trial data. Industry analyses project potential reductions in clinical trial timelines and costs of 20 to 50 percent through faster patient recruitment, adaptive trial designs, real-time safety monitoring, and smarter patient stratification.
Sanofi's application of digital twin simulations in an asthma drug trial illustrates the capacity of artificial intelligence to refine dosing strategies and eliminate unnecessary trial cohorts. By creating virtual patient cohorts and determining that an additional Phase II dose-finding cohort was unnecessary, Sanofi achieved cost savings estimated in the millions of pounds and reduced trial duration by six months. Formation Bio, a biotech company explicitly founded to exploit AI in clinical operations, has demonstrated practical achievement of these projected timelines by automating administrative and analytical tasks around trials. The company reports savings of as much as 50 percent of trial time through automation of patient recruitment, regulatory filings, and data analysis.
Exscientia, a UK-based AI pharma company, piloted the application of AI to patient-drug matching by providing an AI system with tumour samples from 143 cancer patients and a library of 139 cancer drugs that had not been originally matched to those patients. The AI system predicted which drug would work best for each individual tumour, and in treating 56 heavily pre-treated cancer patients with AI-recommended drugs, 54 percent experienced disease control lasting approximately one-third longer than their previous therapies. This result suggests substantial value from AI-driven patient stratification and personalised therapy matching.
The prediction of compound toxicity represents a critical bottleneck in preclinical drug discovery, where compounds demonstrating attractive pharmacodynamic properties may fail development due to off-target toxicity, drug-drug interactions, or cardiotoxicity. Artificial intelligence models trained on historical toxicology data, chemical structures, and mechanism of action information enable researchers to predict adverse safety liabilities early in the discovery process, thereby reducing the number of failed compounds entering more expensive development phases. The integration of AI-driven toxicity prediction with non-animal methods and alternative testing strategies aligns with evolving regulatory requirements, including the FDA Modernisation Act 2.0, which explicitly allows drug developers to use non-animal testing methods including AI-based predictive toxicology.

The market for AI drug discovery platforms spans a diverse ecosystem of technology vendors, platform companies, and service providers offering solutions with varying degrees of specialisation, therapeutic area focus, and stage of development. The following table summarises key platforms operating within or accessible to UK pharmaceutical organisations:
| Platform | UK Presence | Specialisation | Key Metric | Typical Investment |
|---|---|---|---|---|
| Recursion Pharmaceuticals | UK operations; BioHive supercomputers | End-to-end platform; high-throughput biology | 23 petabytes proprietary data; Roche £12B partnership | Enterprise partnership; £500M+ milestone payments |
| Insilico Medicine | European operations; clinical focus | Preclinical acceleration; PandaClaw platform | 12-18 months preclinical (vs. 4.5 years traditional) | £48M milestone partnership demonstrated |
| Isomorphic Labs | London-based; Google DeepMind spinout | Structure prediction; IsoDDE engine | 50% success on novel protein pockets vs. 23.3% AlphaFold 3 | Proprietary licensing; partnership model |
| Exscientia | Oxford-based; UK-founded | Molecular design; patient stratification | 54% disease control in heavily pre-treated cancer patients | Integration into Recursion; partnership licensing |
| BenevolentAI | UK-based; AstraZeneca partnerships | Knowledge graphs; target identification | AstraZeneca collaborations in rare disease; kidney disease | Partnership licensing; enterprise negotiation |
| Schrödinger | Global; UK accessible | Predictive toxicology; molecular modelling | Cardiac toxicity; CYP450 interactions | £50K-200K upfront; 10-20% annual licensing |
Recursion Pharmaceuticals represents an end-to-end platform approach integrating automation, high-throughput biology, and machine learning. The company operates advanced supercomputers and maintains a proprietary dataset spanning more than 23 petabytes, providing the foundation for training AI models capable of predicting biological responses at scale. Insilico Medicine has published performance metrics demonstrating substantial timeline compression, completing preclinical stages in 30 months compared to the traditional 6-year timeline, with preclinical candidates nominated in 12 to 18 months compared to traditional approaches requiring 4.5 years.
Isomorphic Labs, the London-based AI drug discovery company spun out from Google DeepMind, has emerged as a leader in protein structure prediction and biomolecular interaction modelling. The company's proprietary IsoDDE system achieved 50 percent success rate on novel protein pockets and ligands compared to 23.3 percent for AlphaFold 3 on challenging benchmarks. For antibody-antigen interface modelling, IsoDDE achieved 75.58 percent accuracy compared to 47.90 percent for AlphaFold 3, demonstrating capability on mechanistically complex cases where existing systems fail.
The regulatory landscape governing artificial intelligence in pharmaceutical development has evolved substantially between 2024 and 2026, providing clarity for UK pharmaceutical organisations planning integration of AI-discovered or AI-optimised compounds into regulatory submissions.
The Medicines and Healthcare products Regulatory Agency established the National Commission into the Regulation of AI in Healthcare on 26 September 2025, a non-statutory body tasked with reviewing existing regulations and recommending improvements for a new regulatory framework scheduled for publication in 2026. The MHRA's consistent approach emphasises that AI should augment rather than replace expert judgement, that data governance and documentation are essential, and that models must be assessed against their intended context of use with clear accountability and lifecycle management.
On 14 January 2026, the European Medicines Agency and the U.S. Food and Drug Administration agreed to a set of ten common principles for the use of artificial intelligence across the medicines lifecycle, representing unprecedented harmonisation of international regulatory expectations. These principles establish foundational elements including human-centric design, risk-based approach with proportionate validation, adherence to legal and ethical standards, clear context of use and performance specifications, multidisciplinary expertise, appropriate data governance, model design following best practices, risk-based performance assessments, transparency and traceability, and appropriate post-market surveillance. This regulatory harmonisation provides UK organisations with a clear framework within which to operate.
Pharmaceutical organisations implementing AI in regulated operations must validate AI and machine learning systems in accordance with established quality risk management frameworks such as ICH Q9 Quality Risk Management. This approach permits innovation by enabling organisations to demonstrate safety and efficacy through appropriate risk controls rather than prescriptive validation requirements, allowing flexibility in validation strategy whilst maintaining rigorous assurance.
The U.S. Food and Drug Administration has issued numerous non-binding guidance documents addressing considerations specific to AI-enabled pharmaceuticals. FDA Commissioner Dr. Martin Makary stated in January 2026 that the FDA is developing a new regulatory framework for AI that is "smarter" and "more forward thinking," with intention to take a "common sense" approach including a shift toward "deregulatory direction for low-risk products."
The European Union's pharmaceutical regulatory reform, agreed by the European Parliament and Council in December 2025, creates additional possibilities for testing innovative AI-driven methods for medicines in controlled regulatory environments. The new Pharma Package accommodates broader use of AI across the full lifecycle of medicines in regulatory decision-making and mandates the European Medicines Agency to issue non-binding guidance on AI deployment across the entire medicinal product lifecycle. The package also establishes trusted testing environments and data quality accelerators for AI-enabled biotechnology, promoting responsible experimentation and data interoperability.
Pharmaceutical and biotech organisations face critical strategic decisions regarding whether to build AI capabilities in-house, purchase commercial platform solutions, or establish hybrid partnerships combining elements of both approaches. The financial analysis reveals substantial differences in initial capital requirements and ongoing operational costs. Building AI capabilities in-house entails significant upfront investment in salaries for data scientists and engineers (typically USD 200,000 to USD 500,000 annually per senior specialist), computing infrastructure, data curation, and compliance infrastructure. Industry estimates suggest that an in-house AI project requires USD 500,000 to USD 2 million in initial capital plus approximately 30 to 40 percent of that investment annually for ongoing development and maintenance.
In contrast, purchasing commercial AI platforms typically requires upfront licensing fees in the range of USD 50,000 to USD 200,000 and smaller annual subscription costs representing 10 to 20 percent of the upfront investment. For pharmaceutical organisations with well-defined use cases and limited requirement for proprietary customisation, the commercial platform approach often represents substantially lower total cost of ownership, particularly over shorter time horizons. However, for very long-lived projects with anticipated utilisation across multiple therapeutic programmes, the amortised cost of in-house development may ultimately prove lower than perpetual licensing fees.
Successful organisations implementing AI tend to follow differentiated implementation pathways depending on their existing technical capabilities, data infrastructure maturity, and strategic priorities. Early adopters among major pharmaceutical companies have established dedicated artificial intelligence and machine learning centres of excellence with clear governance structures and multidisciplinary teams combining clinical, technical, and data science expertise. These organisations embed AI into previously existing deterministic workflows rather than replacing them entirely, thereby creating hybrid systems that combine AI predictions with traditional safety and compliance frameworks.
Successful AI integration in pharmaceutical development typically follows a phased implementation approach that progressively scales capability whilst managing risk and building internal expertise.
The initial phase focuses on defining strategic positioning, establishing governance structures, and evaluating pilot opportunities. Organisations should establish AI governance committees including regulatory, quality, and scientific experts. Leadership must commit to multi-year investment, and strategic clarity must be developed regarding AI's role within competitive positioning and R&D strategy. During this phase, organisations should identify two to three high-value pilot use-cases with clear success metrics and well-defined problems susceptible to AI solutions.
The second phase prioritises establishment of data governance, standardisation, and infrastructure. Clear data ownership must be established, standardised data formats implemented, data provenance documented, and robust quality control processes created for data collection and curation. Pilot projects should be executed against well-defined use-cases, with performance measured against both technical metrics (model accuracy) and business metrics (timeline compression, cost reduction). Workforce capability development should commence with structured AI training programmes for pilot team members.
The third phase involves validation of pilot outcomes and beginning integration with regulatory and quality management systems. Organisations should define formal performance assessment frameworks, establish context-of-use documentation for candidate AI systems, and develop risk-based validation approaches aligned with ICH Q9 principles. Regulatory engagement should commence with competent authorities to ensure implementation approaches align with emerging regulatory guidance. Successful pilot outcomes should be documented and prepared for broader organisational sharing.
The fourth phase involves expanding AI deployment across additional programmes and therapeutic areas whilst building internal capability. Successful pilot systems should be operationalised, with integration into established quality management systems. Workforce development should accelerate, with structured AI training programmes extended across research and development teams. Organisations should evaluate build-versus-buy decisions systematically, recognising that commercial platforms have matured substantially and often offer compelling value propositions for standard use-cases. Strategic partnerships with established AI drug discovery companies may provide access to advanced capabilities without requiring full internalisation of development risk.
The fifth phase involves enterprise-wide integration of AI capabilities, with established systems integrated into core R&D workflows. Continuous monitoring and optimisation should be embedded in ongoing operations, with model performance tracked against defined specifications and context of use. Post-market surveillance and monitoring approaches should be established for AI-influenced regulatory submissions. Workforce expertise should be sustained through ongoing professional development and certification programmes. The organisation should establish processes for capturing lessons learned and continuous improvement across all AI-enabled operations.
The return on investment analysis for AI implementation in pharmaceutical development requires differentiation between preclinical and clinical phases, as the financial dynamics and timeline impacts differ substantially.
The preclinical phase, extending from target identification through investigational new drug application, represents an established domain where AI demonstrates proven return on investment with documented cost and timeline reductions. Industry analyses project 25 to 50 percent reductions in time and cost for early drug research through preclinical completion, with some estimates placing preclinical cost savings as high as 50 to 80 percent. For full drug development programmes where out-of-pocket costs run approximately USD 2.6 billion per approved drug, AI could reduce that figure by 15 to 67 percent, driven by fewer failed experiments and faster lead optimisation.
Clinical development, spanning Phase I through regulatory approval, represents the highest-cost and longest-duration phase of drug development, with Phase III alone consuming 40 to 50 percent of total clinical development budgets. Analyses project potential 20 to 50 percent reductions in clinical trial timelines and costs through faster recruitment, adaptive trial designs, real-time safety monitoring, and smarter patient stratification. The potential industry-wide savings from AI-driven clinical development optimisation have been estimated at USD 20 to USD 30 billion annually by 2030.
The most important insight from examining both preclinical and clinical development together is that return on investment compounds across the development pipeline. An AI-optimised lead that enters Phase I with superior absorption, distribution, metabolism, excretion, and toxicity properties is less likely to fail in Phase II. A Phase II optimised patient stratification strategy produces cleaner efficacy signals, reducing Phase III sample size requirements. Each stage's efficiency gain feeds the next, creating multiplicative benefits rather than simple additive ones. Pharmaceutical companies integrating AI successfully across their full pipelines could potentially achieve cost savings of USD 390 million to USD 1.7 billion per approved drug, representing transformative financial impact that justifies substantial upfront investment in data infrastructure, talent acquisition, and organisational change management.
Successful AI implementation in pharmaceutical development requires substantial investment in workforce capability development, as the market supply of specialists combining drug development domain expertise with advanced artificial intelligence and machine learning skills remains substantially constrained. The pharmaceutical industry faces substantial deficiencies in workforce skills representing a critical barrier to successful AI adoption across the development pipeline. Effective AI drug discovery requires "bilingual" professionals—experts fluent in both biomedicine and artificial intelligence—where training such talent may take longer than developing the software systems themselves.
The regulatory landscape is increasingly mandating structured, certified AI training programmes as a strategic necessity rather than optional professional development. The EU AI Act's high-risk provisions take effect on August 2, 2026, creating new compliance requirements for AI used in drug development and clinical settings. The FDA's 2025 draft guidance on AI in drug development makes clear the expectation that organisations will have trained personnel capable of managing, validating, and operating AI systems correctly in regulated environments. Pharmaceutical organisations should invest substantially in workforce development, recognising that AI's value depends on widespread capability for appropriate application and interpretation. Structured, certified AI training programmes should be prioritised, with particular focus on developing "bilingual" professionals fluent in both domain expertise and machine learning.
Despite substantial opportunities, multiple significant challenges constrain effective AI adoption across UK pharmaceutical organisations.
The most fundamental challenge constraining effective AI implementation is data quality and fragmentation across pharmaceutical and biotech organisations. Molecular databases, clinical records, and real-world evidence are often stored on disparate platforms with inconsistent formatting, incomplete documentation, and limited interoperability. This data fragmentation impedes the data-driven decision-making necessary for effective AI application. Data quality matters more than model complexity in determining actual AI system performance in pharmaceutical applications.
Artificial intelligence systems employed in pharmaceutical development often face tension between predictive performance and regulatory-grade assurance. Deep learning models with clinical potential are frequently challenging to justify in regulator-ready form because of complex data dependencies, limited interpretability, and performance sensitivity to distribution shift. The MHRA's consistent position that "AI should be used to augment expert judgement, not replace it; that data governance, documentation and validation are essential; and that models must be assessed against their intended context of use" establishes the non-negotiable framework within which AI systems must operate.
Many pharmaceutical organisations remain stuck in pilot mode, unable to scale AI across global operations whilst managing regulatory complexity and patient data sensitivity. The transition from proof-of-concept pilots to production-grade, enterprise-scale implementations requires substantial organisational change management, establishment of AI governance committees, integration of AI outputs into established quality management systems, and retraining of existing workforce. The volume of data generated in modern clinical trials has reached an inflection point that traditional systems can no longer manage: in 2012, a Phase III trial collected approximately 900,000 data points, whereas in 2025, that number has expanded to approximately 6 million.
Based on contemporary evidence regarding AI adoption in pharmaceutical development, UK organisations should consider the following strategic recommendations:
First, establish strategic clarity and governance structures. Organisations should prioritise development of strategic clarity regarding AI's role within their competitive positioning and R&D strategy, recognising that AI represents a productivity multiplier and talent enabler rather than a solution for all pharmaceutical development challenges. Leadership commitment to multi-year investment in AI capability, including substantial investment in workforce development, data infrastructure, and governance frameworks, is prerequisite for successful enterprise-scale implementation. Organisations should establish dedicated AI and machine learning centres of excellence with clear governance structures and multidisciplinary teams.
Second, prioritise data infrastructure investment. Pharmaceutical organisations should prioritise investment in data governance, standardisation, and infrastructure before initiating AI model development. Establishing clear data ownership, implementing standardised data formats, documenting data provenance, and creating robust quality control processes are essential prerequisites. Organisations should evaluate whether commercial cloud platforms offering managed data services might provide more efficient and scalable solutions than building custom infrastructure.
Third, engage proactively with regulatory frameworks. UK organisations should engage proactively with MHRA guidance and regulatory frameworks, recognising that the regulatory environment is evolving but established on principles of human-centric design, risk-based approach, clear context of use, and proportionate validation. Organisations should establish AI governance committees including regulatory, quality, and scientific experts to oversee AI implementation and ensure alignment with emerging regulatory expectations. Implementation of frameworks such as ICH Q9 risk-based approaches to AI validation will facilitate regulatory acceptance.
Fourth, invest in workforce capability development. Pharmaceutical organisations should invest substantially in workforce development, recognising that AI's value depends on widespread capability for appropriate application and interpretation. Structured, certified AI training programmes should be prioritised, with particular focus on developing professionals fluent in both domain expertise and machine learning. Organisations should consider partnerships with academic institutions, specialist training providers, and established technology companies to accelerate capability development.
Fifth, evaluate build-versus-buy-versus-partner decisions systematically. Organisations should recognise that commercial platforms have matured substantially and offer compelling value propositions for standard use-cases. For organisations with limited proprietary AI requirements and substantial internal data science skill constraints, commercial platform licensing often offers substantially lower total cost of ownership than in-house development. For larger organisations with multiple therapeutic programmes, hybrid approaches combining commercial platforms with internal customisation may provide optimal balance. Strategic partnerships with established AI drug discovery companies may offer access to advanced capabilities without requiring full internalisation of development risk.
Organisations seeking to understand AI implementation in healthcare contexts can benefit from examining comprehensive AI for healthcare frameworks, which provide foundational governance and implementation patterns applicable across pharmaceutical operations. Similarly, understanding AI implementation guidance developed for broader healthcare contexts helps pharmaceutical organisations navigate similar challenges around data governance, regulatory alignment, and workforce capability development.
The financial implications of AI implementation extend beyond drug discovery into supporting functions. Financial operations powered by AI enable pharmaceutical companies to manage the complex cost accounting associated with multi-year development programmes, whilst regulatory reporting automation supports compliance with evolving reporting requirements surrounding AI-discovered drugs. Understanding strategic AI consulting approaches helps pharmaceutical leadership make informed build-versus-buy decisions aligned with competitive positioning.
Governance and risk management frameworks become increasingly critical as AI systems influence regulatory submissions and patient outcomes. Comprehensive AI governance frameworks provide foundational structures within which pharmaceutical organisations can embed AI into existing quality management systems. For organisations managing complex multi-disciplinary teams around AI implementation, risk management approaches specifically designed for artificial intelligence help identify and mitigate model drift, algorithmic bias, and cybersecurity vulnerabilities characteristic of high-risk AI applications in healthcare.
Broader operational transformation enabled by AI extends into core business processes. AI-driven business process automation demonstrates how organisations compress timelines and reduce costs through intelligent automation of regulatory filings, trial administration, and data management tasks—capabilities directly applicable to pharmaceutical development operations where Formation Bio has already demonstrated 50 percent trial timeline reduction through similar approaches.
For healthcare-specific applications of AI beyond drug discovery, organisations should consider related focused articles including AI for mental health applications, AI-powered medical imaging, AI applications in dentistry, pharmacy operations powered by AI, healthcare compliance frameworks, and AI-enhanced clinical documentation systems. Each of these specialised applications provides learnings applicable to pharmaceutical development contexts.
Published evidence indicates timeline compression of 12 to 18 months for preclinical candidate nomination compared to the traditional 4.5-year approach (Insilico Medicine), 30 percent reduction in early-stage development (Merck KERMT model), and 20 to 50 percent reduction in clinical trial timelines through optimised patient recruitment and adaptive designs. Sanofi demonstrated six-month reduction in asthma trial duration through digital twin optimisation. The magnitude of timeline compression varies by therapeutic area, development phase, and AI application specificity, with highest compression rates documented in preclinical discovery and clinical trial optimisation phases.
The primary barriers include data quality and fragmentation across existing systems, insufficient workforce expertise in AI and machine learning, absence of robust data governance frameworks, regulatory uncertainty (though substantially clarified in 2025-2026), organisational change management challenges, and limited integration of AI outputs into established quality management systems. Many organisations lack clear governance structures defining accountability and decision-making authority for AI-influenced decisions. Model explainability and regulatory-grade assurance remain challenging for complex deep learning models, creating tension between predictive performance and regulatory acceptability.
The decision depends on organisational size, existing data science capability, strategic importance of proprietary differentiation, and anticipated scope of AI application across multiple programmes. Commercial platforms offer substantially lower total cost of ownership (USD 50,000-200,000 upfront plus 10-20 percent annually) compared to in-house development (USD 500,000-2 million upfront plus 30-40 percent annually). For organisations with well-defined use-cases and limited proprietary requirements, commercial platforms typically provide better value. For large organisations with multiple programmes and substantial existing data science capacity, hybrid approaches combining commercial platforms with targeted internal development often provide optimal balance. Strategic partnerships with established AI drug discovery companies offer an alternative pathway to advanced capability access without full internalisation of development risk.
Regulatory authorities including the MHRA, EMA, and FDA have established that AI-discovered candidates should be evaluated using the same rigorous standards as traditionally discovered drugs, with evaluation focused on data provenance, model validation, performance assessment across intended context of use, and ongoing monitoring. The primary regulatory requirement is not that drugs be discovered through traditional methods, but rather that sponsors provide robust evidence of safety and efficacy regardless of discovery method. The MHRA established that AI should augment rather than replace expert judgement, that clear documentation of data governance and model validation is essential, and that models must be assessed against their intended context of use with clear accountability and lifecycle management. Regulatory science initiatives continue to explore how AI can strengthen evidence generation whilst maintaining robust assurance.
AI demonstrates clear value in four primary clinical development applications: protocol optimisation through virtual patient cohorts (Sanofi's digital twin approach saved six months and millions of pounds in asthma trial), patient recruitment automation (Formation Bio demonstrated 50 percent timeline reduction through automated patient identification and recruitment), adaptive trial design with real-time safety monitoring (enabling dynamic trial structure adjustments based on emerging efficacy and safety data), and patient stratification (Exscientia's cancer patient-drug matching achieved 54 percent disease control rate approximately one-third longer than previous therapies in heavily pre-treated populations). The highest-impact applications focus on the highest-cost phases of development, where even modest timeline compression generates substantial financial benefit and accelerates patient access to new treatments.
Return on investment timelines vary substantially by therapeutic area and development phase. Preclinical applications demonstrate rapid return through cost savings and timeline compression, with published evidence indicating cost reductions of 25 to 80 percent and timeline reduction from 6 years to 30 months (Insilico Medicine). However, realisation of full pipeline benefits requires multi-year investment in data infrastructure, workforce capability, governance frameworks, and organisational integration. Most pharmaceutical organisations should anticipate 18 to 24 months to establish foundational infrastructure, execute initial pilots, and begin scaled deployment, with full enterprise integration requiring 24 to 36 months or longer depending on organisational size and complexity. The compounding effect across the entire development pipeline—where each stage's efficiency gain increases the likelihood of success in subsequent phases—suggests that organisations committed to full-pipeline AI integration could realise cost savings of USD 390 million to USD 1.7 billion per approved drug, making the investment case compelling despite substantial upfront costs.
Artificial intelligence technologies have evolved substantially between 2024 and 2026 from speculative promises and limited proof-of-concept demonstrations to production-grade systems demonstrating quantifiable acceleration of drug discovery timelines, improved candidate quality, and reduced development costs across multiple therapeutic modalities. The UK biotechnology market, valued at USD 22.00 billion in 2024, is positioned to expand substantially as AI-driven innovation becomes embedded within research and development operations across the pharmaceutical sector. The regulatory environment has stabilised around principles emphasising human-centric design, risk-based validation, clear context of use, and proportionate governance—frameworks that provide both flexibility for innovation and assurance of safety and efficacy.
For UK pharma and biotech leaders, the evidence is compelling: AI-discovered candidates are advancing through development with superior conversion rates, timeline compression is documented across preclinical and clinical phases, and the financial case for investment is robust with potential cost reductions of USD 390 million to USD 1.7 billion per approved drug. However, realisation of these benefits depends critically upon successful navigation of substantial implementation challenges. Data quality, workforce capability development, regulatory alignment, and organisational change management remain significant barriers constraining full-scale adoption across the UK pharmaceutical sector.
The most advanced adopters have recognised that AI succeeds as a tool augmenting expert judgement within carefully designed workflows, not as an autonomous decision-making system replacing human scientific expertise and regulatory oversight. Strategic pharmaceutical leaders should approach AI implementation as a multi-year transformation requiring substantial investment in data infrastructure, talent acquisition, governance frameworks, and organisational change management. The UK government's strategic positioning of AI adoption as a priority for the life sciences sector, combined with substantial funding commitments through UKRI and the VPAG Investment Programme, creates favourable conditions for pharmaceutical innovation.
For UK pharmaceutical and biotech leaders seeking to maintain competitive positioning within the rapidly evolving global drug discovery landscape, investment in AI capability is no longer discretionary but rather fundamental to long-term strategic competitiveness. The time for pilot projects and cautious evaluation has passed. The organisations that will lead the next decade of pharmaceutical innovation will be those that commit decisively to AI integration, establish robust governance and validation frameworks aligned with regulatory expectations, invest substantially in workforce capability, and embed AI as a foundational layer within their research and development operations.
Helium42 specialises in helping pharmaceutical and biotech organisations navigate AI implementation strategically. Whether you are evaluating build-versus-buy decisions, establishing governance frameworks, developing workforce capability, or scaling pilot programmes to enterprise deployment, our AI consultancy team combines deep pharmaceutical domain expertise with contemporary AI and machine learning knowledge.
We help UK pharma companies:

The AI Software Development Market in 2026 £337.75bn UK AI market size by 2032 (26.4% CAGR) £800–£1,500+ Daily rates for mid-market...

Clinical Documentation as a Crisis: The Hidden Cost of Manual Note-Taking The NHS faces one of its most persistent operational challenges:...

Healthcare Compliance: The Regulatory Landscape Transformed by Artificial Intelligence UK healthcare organisations operate within one of the world's...